Welcome to WordPress. This is your first post. Edit or delete it, then start writing!
OPINCHARGE Scientific Publication: Electrochemical removal of HF from Li-ion battery electrolytes
In the OPINCHARGE Scientific Publications series, we highlight research advancing the understanding and optimisation of battery materials and processes.
This study explores a novel electrochemical approach to remove hydrofluoric acid (HF) impurities from carbonate-based lithium-ion battery electrolytes.
The publication “Electrochemical Removal of HF from Carbonate-based LiPF6-containing Li-ion Battery Electrolytes” was authored by Xiaokun Ge, Marten Huck, Andreas Kuhlmann, Hans-Georg Steinrück (Forschungszentrum Jülich), Michael Tiemann, Christian Weinberger, Xiaodan Xu and Zhenyu Zhao (Paderborn University).
The authors are affiliated with the Forschungszentrum Jülich and Paderborn University, both active in advanced electrochemistry and battery research.
Read the publication: https://zenodo.org/records/14974741

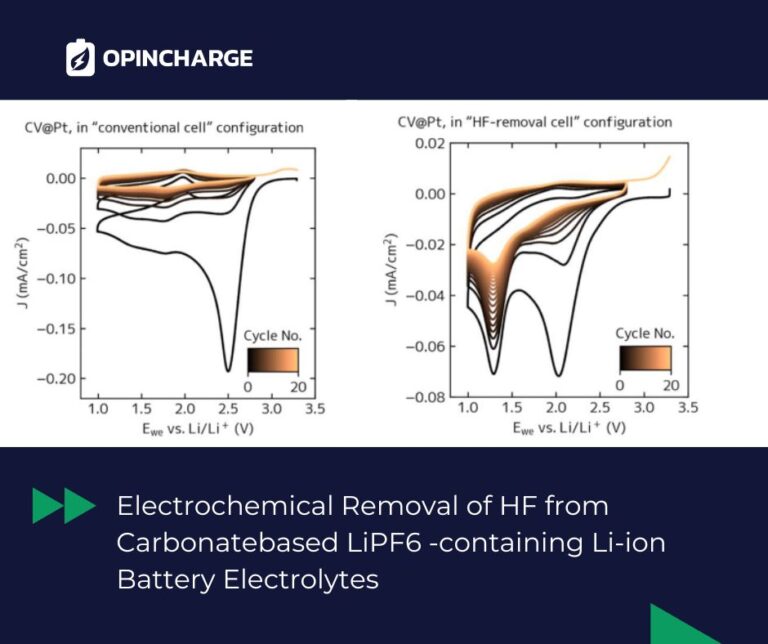
Why HF matters in battery electrolytes
Lithium-ion battery electrolytes typically contain LiPF6 dissolved in carbonate solvents. Due to chemical instability and trace water content, small amounts of hydrofluoric acid (HF) are almost always present. Even at low concentrations, HF can significantly affect battery behaviour.
It influences solid electrolyte interphase (SEI) formation, contributes to cathode degradation, and complicates the study of fundamental electrochemical processes. Controlling or removing HF is therefore essential for both battery performance and research accuracy.
A new electrochemical approach
The study introduces a simple and efficient electrochemical method to selectively remove HF from battery electrolytes. The concept is based on applying a suitable potential to a high surface-area metallic electrode.
Under these conditions, HF reacts preferentially while the other electrolyte components remain largely unaffected. This enables targeted removal of HF without altering the overall electrolyte composition.
Key findings
The researchers demonstrate that:
- HF can be selectively removed from LiPF6-based electrolytes using an electrochemical approach
- the method works effectively in common electrolyte systems such as LP40 and LP30
- the process enables better control of interfacial chemistry and electrochemical measurements
The results confirm that HF is the most electrochemically unstable component in these systems and can therefore be removed without disrupting other species.
Supporting better battery research and design
Removing HF from electrolytes opens new opportunities for battery research. It allows scientists to study interfacial processes more precisely, including direct anion reduction and SEI formation.
In the long term, such insights can support the development of more stable, efficient and longer-lasting lithium-ion batteries. The study also suggests that this approach could be scaled up for electrolyte purification processes, contributing to improved battery manufacturing and performance.